
mucoderm®
mucoderm®
Mucoderm is a stable three-dimensional matrix of acellular porcine collagen that offers a safe alternative to autologous soft tissue transplants. The production process provides a type I/III collagen matrix with a similar structure to connective tissue.
Why mucoderm®?
1
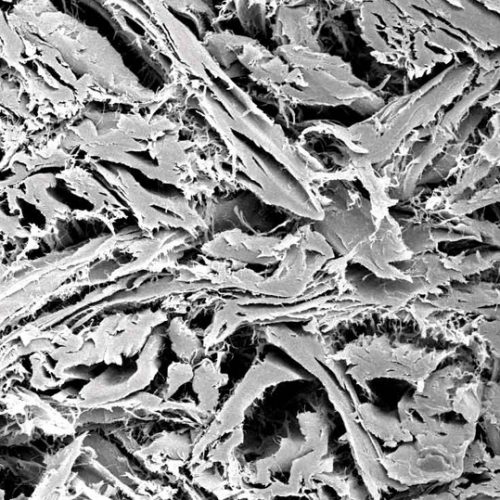
Natural structure of porcine type I/III collagen
Mucoderm is a collagen matrix that maintains the natural properties of the original tissue throughout the production process. Furthermore, it is an easy-to-handle membrane with tear resistance and dimensional stability. The preservation of the natural structure is also the reason why mucoderm is similar to the human dermis, making it a suitable alternative to autologous connective tissue gingival grafts.
2
High level of safety
The cleaning process to which mucoderm is subjected effectively removes all non-collagenous cells and proteins, possible immunogens, bacteria and viruses. Mucoderm is a regulated medical device under EC guidelines. Its manufacture is subject to a rigorous quality control system based on international standards and is regularly audited by the notified body and the authorities.
3
Integration, revascularisation and degradation
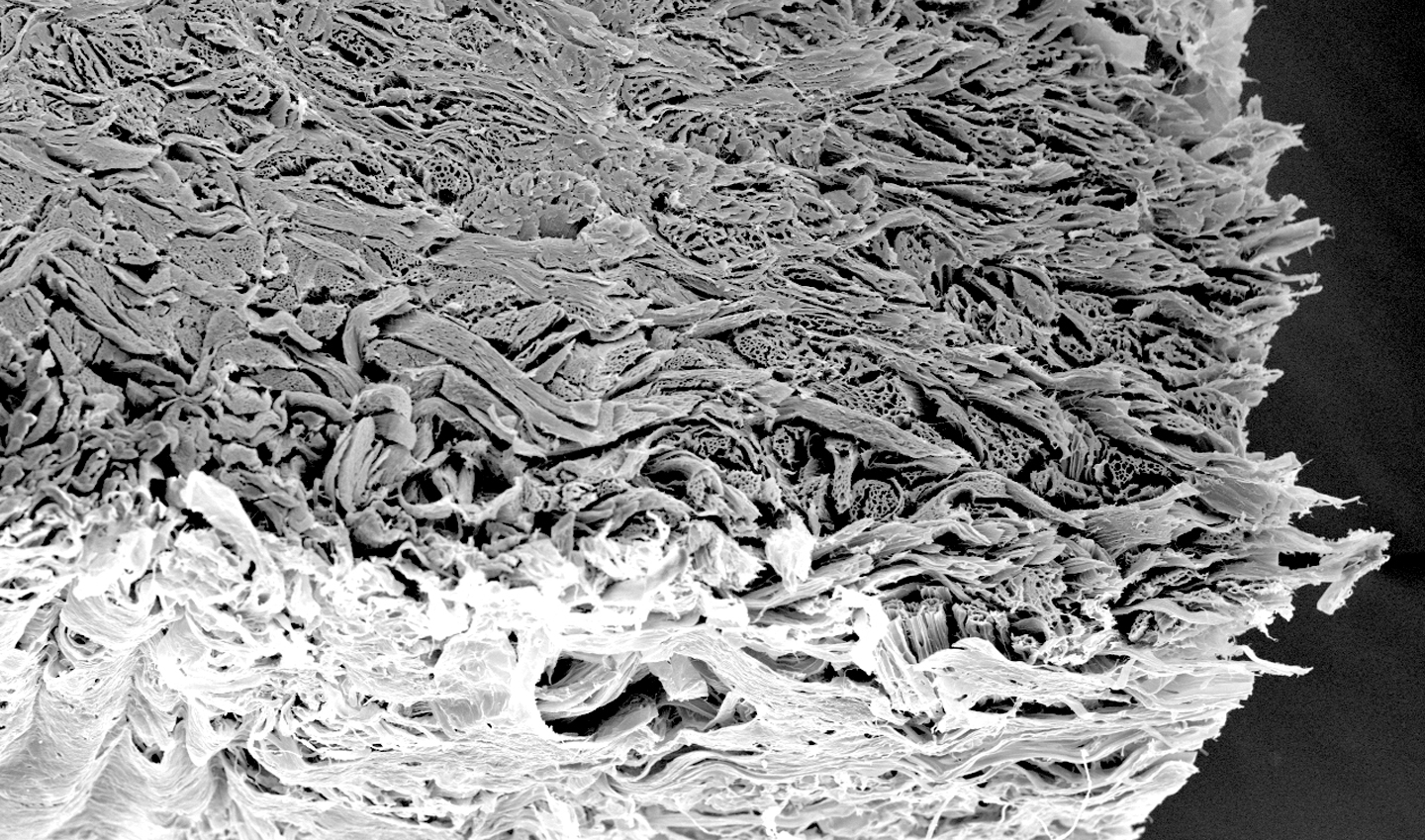
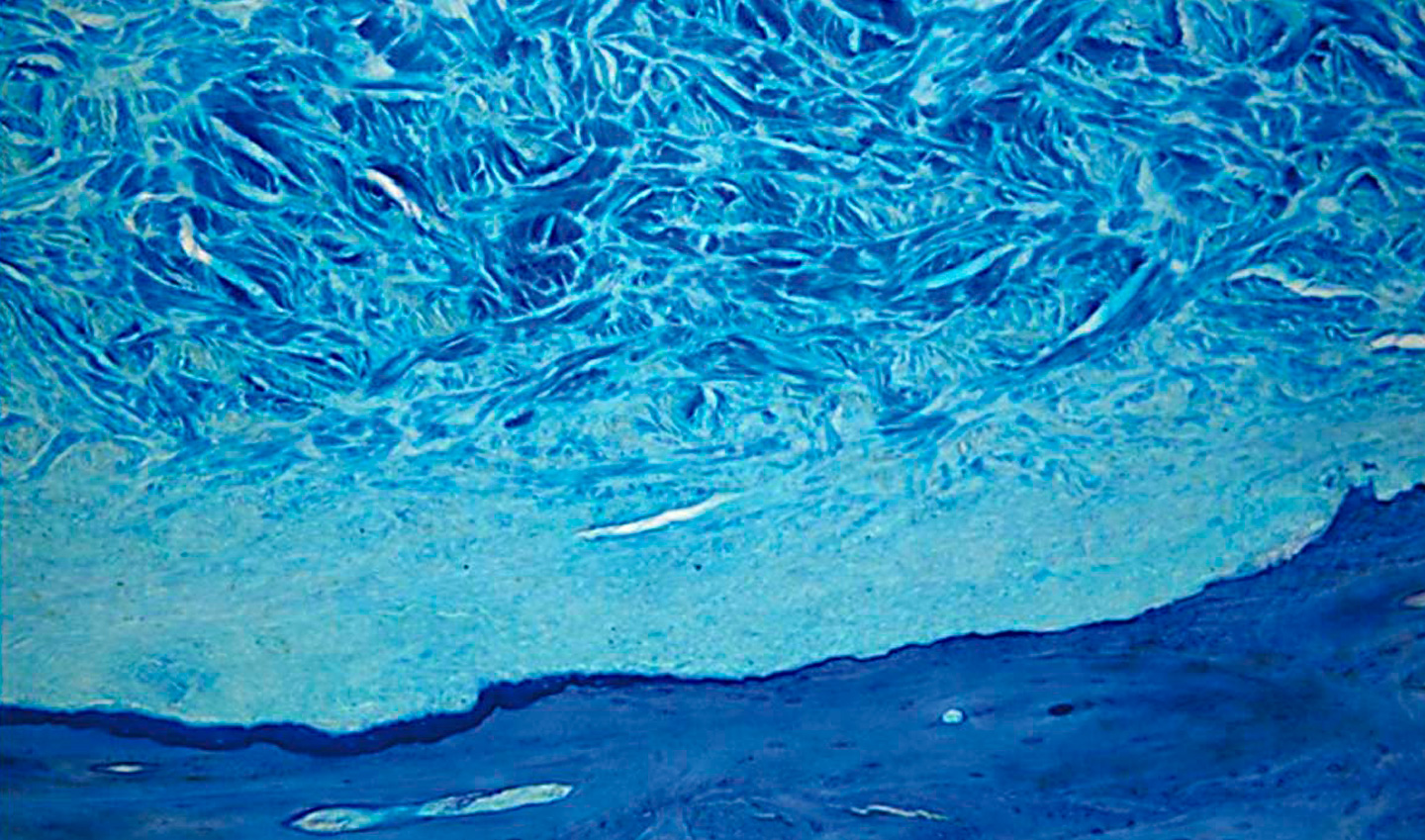
Mucoderm is characterised by an open porous collagen structure. This unique structure makes it an ideal scaffold for cell and blood vessel growth, and promotes rapid integration and revascularization of tissues.
In the 4 to 8 weeks following implantation, a continuous degradation of the membrane can be observed, as well as an increase in homogeneous cell distribution. After 8 weeks, 20% of the volume of the original matrix is still present, favouring the formation and reorganisation of the connective tissue. After 12 weeks, the mucoderm membrane is almost completely replaced by newly formed tissue.
4
Membrane exposure
The mucoderm matrix can be partially exposed as long as revitalisation of the region surrounding or underlying the area that houses the membrane is guaranteed. The exposure of the matrix is also acceptable in the case of a vestibuloplasty, taking into account that the membrane has to be sutured to the periosteum. Membrane exposure may be accepted if it is partial and of a small percentage of the total surface area.
5
Hydration protocol
Rehydration should be performed in a sterile saline solution or in the patient's blood for a period of 5 to 20 minutes prior to placement. The time will vary according to the surgical technique to be used and the desired flexibility of the matrix. The flexibility of mucoderm graft increases with hydration time.
6
Indications
- Soft tissue thickening.
- Closure of extraction sockets.
- Substitute for autologous connective tissue in connective tissue grafting techniques.
- Gingival recession coverage.
- Keratinized gum increase.
*Botiss biomaterials products distributed by Klockner S.A. in Spain and Portugal